Nanoknife Focal Therapy for Prostate Cancer
28 November 2023 2026-04-04 2:41Nanoknife Focal Therapy for Prostate Cancer
Do you have Prostate Cancer?

Focal Therapy for Prostate Cancer
A minimally invasive therapy that uses electricity to destroy prostate tumor while preserving urinary and sexual function¹⁻³
• Performed over 200 NanoKnife® cases, one of the highest single-surgeon volumes of NanoKnife® procedures in the world.
• Dr. Jonathan Chan was awarded the Global Excellence in Surgical Innovation Award by AngioDynamics, the manufacturer of Nanoknife (AngioDynamics International LIFE Symposium in Malta, March 2026).
• We are the first and only clinic in Canada to offer Exactvu™ Microultrasound - MRI Fusion NanoKnife® Focal Therapy.⁴
• Offers our patients a 300% improvement in real time imaging resolution over conventional ultrasound used in standard NanoKnife® protocols.⁴
• Designated as a both Consultant & North American Clinical Training Site for AngioDynamics (Manufacturer of NanoKnife®).
Our Approach to Nanoknife
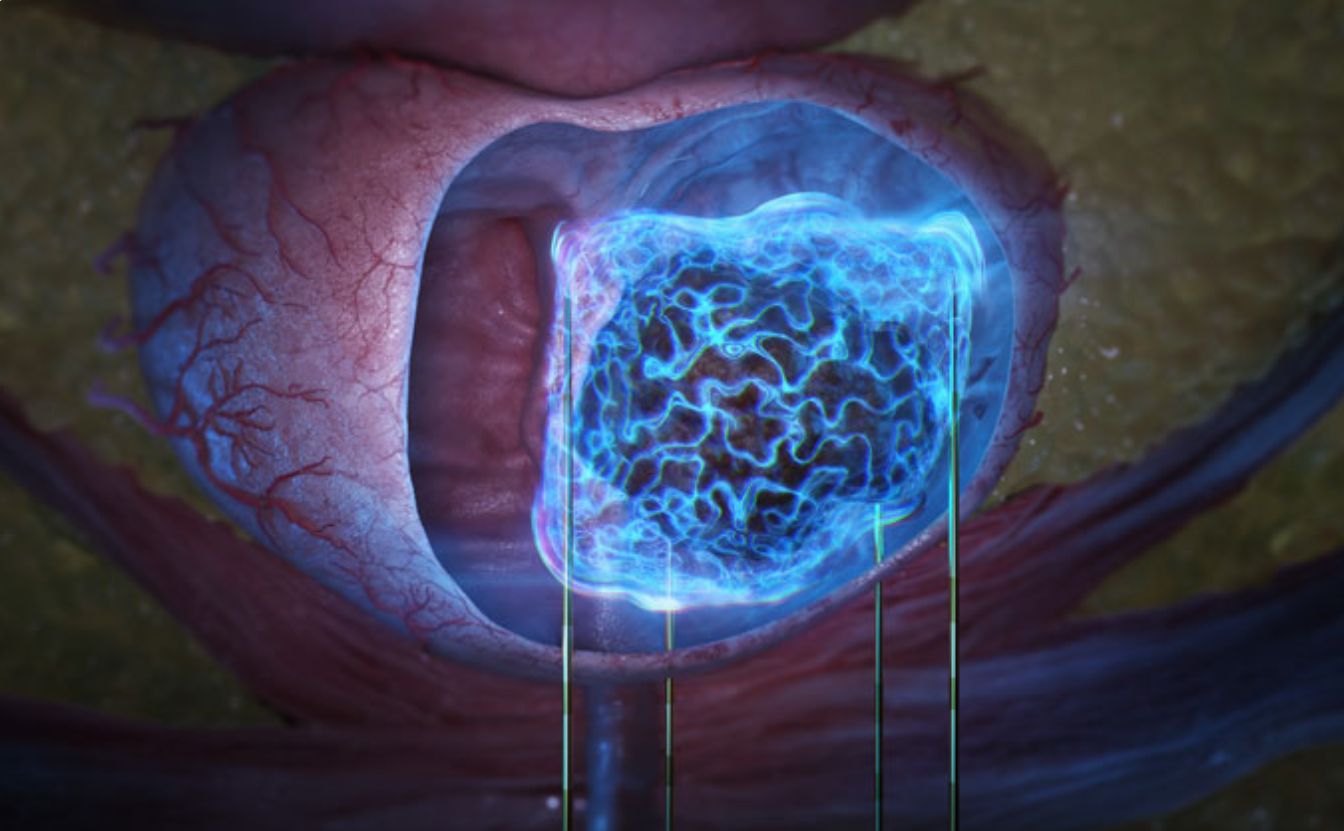
- NanoKnife®, also known as Irreversible Electroporation (IRE), is a breakthrough non-thermal focal treatment for localized prostate cancer.¹ Compared to traditional “whole gland” treatments like surgery or radiation, NanoKnife® targets only the tumor.
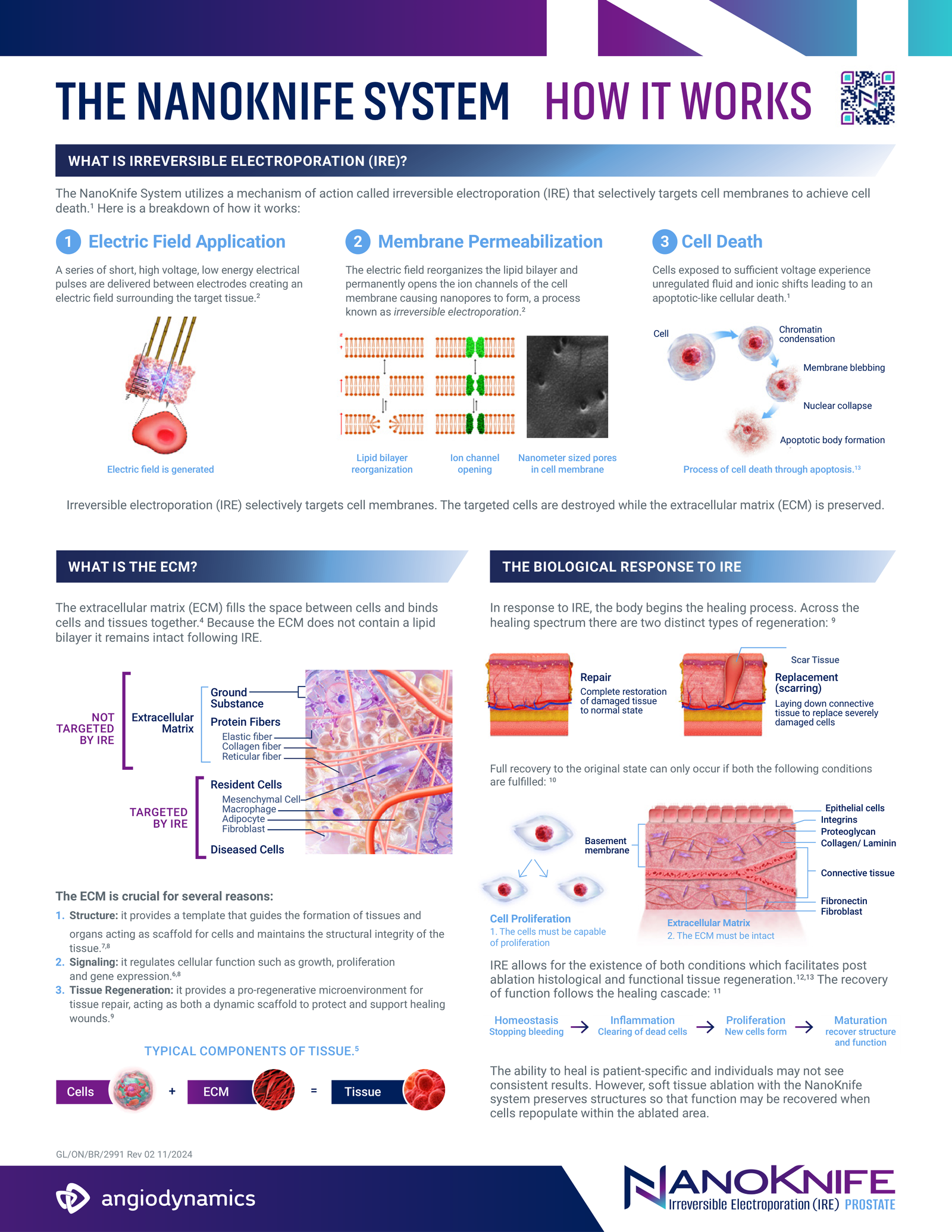
- How NanoKnife® works: Short high voltage electrical pulses are administered through exact placement of electrodes placed around the tumor. The electrical field creates microscopic “nanopores” in the cell membranes of cancer cells.⁴ This process, called electroporation, triggers "programmed cell death" (apoptosis) without using heat or extreme cold which can destroy surrounding healthy tissue.²
- The Non-Thermal Advantage: Nerves, urethra, and sphincter are spared thus improving the likelihood of preserving continence and erectile potency.³ NanoKnife® provides the unique benefit of preserving critical structures which are potentially damaged or removed by traditional treatments (ie surgery or radiation).³
The ATRIUM Urology Advantage:
Why Imaging Matters
- The success of NanoKnife® relies on the surgeon’s ability to "see" the tumor. Standard NanoKnife® protocols use conventional ultrasound which operates at 6-9 MHz. At this lower resolution, many clinically significant prostate cancers remain invisible, leading to approximate needle placement dependent on software mapping or robotic systems.⁴
- ATRIUM Urology offers the world’s highest-resolution imaging - the 29 MHz Exactvu™ Microultrasound - fused with multiparametric MRI (mpMRI). This "dual-vision" approach provides a 300% improvement in resolution allowing for a level of precision previously unattainable.⁴
- Real-time visualization of the prostate’s cellular architecture enables our surgeons to see, target, and destroy the “index lesion” with sub-millimeter accuracy and unprecedented clarity - ultimately preserving your quality of life.
What We Offer You With Nanoknife Focal Therapy for Prostate Cancer
Highest resolution Real-time Microultrasound-MRI fusion Imaging Guidance
Only provider in Canada to integrate high definition 29 MHz Exactvu™ Microultrasound with NanoKnife® (IRE) for the treatment of prostate cancer.⁴ Our protocol visualizes suspicious lesions in real time that are invisible to conventional 6-9 MHz conventional ultrasound.⁴ NanoKnife® electrodes are positioned with sub-millimeter accuracy, effectively targeting and destroying the cancer while sparing the critical nerves and structures responsible for urinary and sexual function.² ³
Reference¹⁵
Multi-disciplinary team of specialist experts
With our dedicated NanoKnife® coordinator, Urologic surgeons, MRI Radiologists, Internal Medicine Specialist, and Anesthesiologist team to ensure a comprehensive patient treatment plan. Committed to the highest standard of patient safety and quality care.
Personalized continuum of care
Offering focal therapy for localized prostate cancer, surgery, and radiation treatment. Offering biomarker testing, transperineal Exactvu™ Microultrasound - MRI fusion prostate biopsy within 1-2 weeks, and expedited MRI and PSMA PET scan if indicated.⁴ We also offer concierge programs for out of province and international patients.
What Is Nanoknife?
What are the Key Benefits of NanoKnife® for Prostate Cancer?
- Ultra-Precision: Targeted electrical fields spare the “critical trifecta:” Nerves, Urethra, and Sphincter.²
- Preservation of Quality of Life: 96% of patients maintain urinary continence and 84% maintain erectile function at 12 months (PRESERVE data).⁵
- Quick Recovery: Outpatient procedure without surgical incisions or hospital stay required. Return to activity within 48–72 hours.³
- Does not preclude other future treatment options: Non-thermal ablation does not cause tissue scarring
thereby keeping all lines of treatment open.¹
The U.S. Food and Drug Administration (FDA) granted 510(k) clearance to the NanoKnife®© System for prostate tissue ablation, specifically targeting intermediate-risk prostate cancer. This approval was based on the pivotal PRESERVE clinical study.⁵
How does Nanoknife treatment work?
The Exactvu™ Microultrasound – MRI Fusion Protocol
By combining the sensitivity of MRI with the real-time resolution of 29 MHz Exactvu™ Microultrasound, we offer the “best-of-both-worlds” approach:⁴
- MRI Planning: We use your mpMRI to map the exact location and boundaries of the tumor.⁴
- Exactvu™ Microultrasound MRI Fusion:By fusing this high-definition live feed with your pre-operative mpMRI (FusionVu™), we ensure NanoKnife® electrodes are placed and bracket the tumor with absolute certainty, ensuring negative margins.⁴,⁵
- Active Targeting:Electrodes are positioned in real-time to account for any movement, ensuring the NanoKnife® treatment zone perfectly encapsulates the cancer.⁵
- Near-Histologic Clarity:While conventional ultrasound has a resolution of ~200–500 microns, our 29 MHz Exactvu™ Microultrasound reaches 70 microns.⁴ This allows us to visualize the micro-architecture of the prostate – specifically the ductal anatomy where cancer begins.⁴
- The PRI-MUS™ Protocol:We utilize the evidence-based Prostate Risk Identification using Exactvu™ Microultrasound (PRI-MUS™) protocol.⁶ This allows our team to identify “index lesions” in real-time that are missed by both standard ultrasound and even some MRI scans.
- Destroy prostate cancer lesion with NanoKnife® (IRE) energy administered through the NanoKnife® 3.0 Generator.⁵
Risks of Nanoknife
- A catheter will be required for a few days post procedure.¹⁻³
- Some men may experience decreased or absent ejaculatory fluid.¹⁻³
- Small amount of blood or seminal fluid in urine from treatment effect.¹⁻³
What to Expect with Nanoknife Focal Therapy for Prostate Cancer
Before the Procedure:
A detailed specialist consultation to assess possible prostate cancer treatment options and eligibility for focal therapy.
Assessment may include prostate cancer biomarkers, multiparametric prostate MRI, and Microultrasound ExactVu™ – MRI fusion transperineal target prostate biopsy if indicated.⁴
During the Procedure:
The procedure is performed under a general anaesthetic and will last approximately 60 minutes.¹⁻³ Four to six electrodes are placed through the skin behind the scrotum (the perineum) into the prostate under ultrasound guidance.¹⁻³,⁵
A short pulse electrical current is then passed sequentially between each of the electrodes enabling the area of prostate mapped out to be destroyed.¹,⁵
After the Procedure:
After treatment the patient will be observed in the day surgery unit before discharged home with a catheter in place.
Postoperatively pain is minimal and patients are discharged with antibiotics, and medications for pain control and bladder spasms as required.¹
A catheter is left for two to five days depending on the extent of the treatment.¹,³
- Long term follow-up monitoring post Nanoknife will include PSA testing, repeat MRI prostate, and prostate biopsy.¹
Scientific Evidence:
Why Our Technology is the 2026 Gold Standard
Our commitment to Exactvu™ Microultrasound is backed by the latest peer-reviewed research, proving that 29 MHz imaging is a game-changer for prostate cancer management.⁴,⁶
- Accuracy Comparable to (or Better Than) MRI
The landmark OPTIMUM Trial (2025), a multicenter randomized study, demonstrated that 29 MHz Exactvu™ Microultrasound is non-inferior to mpMRI in detecting clinically significant prostate cancer.⁷ Notably, Exactvu™ Microultrasound identified significant lesions in 13% of cases where MRI returned a "false negative."⁷
Real-Time Visualization of the "Index Lesion"
Research published in the Journal of Urology (Ghai et al.) and the Canadian Urological Association Journal confirms that the high resolution of 29 MHz imaging allows for "active targeting."⁴,⁹ This means that unlike other NanoKnife® providers who treat a "predicted" area, Atrium Urology treats the visible tumor.
Preservation of Vital Structures
NanoKnife® is non-thermal, but its safety is only as good as its guidance. A study by Lugnani et al. (2022) highlighted that high-resolution guidance is critical for protecting the neurovascular bundles.⁸ Because Atrium Urology can see the nerves and the tumor simultaneously at 29 MHz, we achieve superior outcomes in maintaining erectile function and urinary continence.²,⁸

Leading Clinical Evidence (2025-2026)
The Stricker Series: Long-Term Reliability³ Research led by Dr. Phillip Stricker has been foundational in proving the efficacy of IRE. His extensive series demonstrated³,¹⁰:
- 97% In-Field Clearance: Exceptional success in destroying the targeted cancer.
- Out-field Management: Only a 10% to 12% chance of recurrence in the untreated areas of the prostate, which remains a manageable "out-field" risk.
Prostate Cancer Facts:
Learn more about prostate cancer here:
https://www.procure.ca/en/prostate-cancer/
Frequently Asked Questions and Answers
Is NanoKnife® covered by OHIP?
While some diagnostics are covered, NanoKnife® is currently a private-pay option or may be covered by private insurance. We recommend that you contact your respective insurance provider to confirm coverage details. In most cases, private procedures are eligible for a tax credit. We suggest speaking with your accountant and/or reviewing the CRA guidelines (Medical Expenses Tax Credit).
How does NanoKnife® compare to HIFU?
NanoKnife® is non-thermal, meaning it does not use heat. This reduces the risk of damaging nerves or the “heat sink” effect near blood vessels, making it safer for tumors near critical structures.¹,²
Can the procedure be repeated?
A unique benefit of NanoKnife® is that it does not preclude future surgery or radiation if required.¹,³
What are the risks?
Common temporary side effects include a small amount of blood in the urine, urinary urgency, or decreased ejaculatory fluid.¹,³
Is Atrium Urology the only place in Canada with Exactvu™ Microultrasound NanoKnife®?
Yes. Atrium Urology Associates is currently the only facility in Canada to integrate 29 MHz ExactVu™ Exactvu™ Microultrasound with NanoKnife® (IRE) for the treatment of prostate cancer.
Why is 29 MHz better than the ultrasound used at other clinics?
Most clinics use 6-9 MHz ultrasound, which cannot see the cancer. The 29 MHz Exactvu™ Microultrasound used at Atrium Urology provides 300% better resolution, allowing for real-time visualization of the tumor and precise needle placement to avoid healthy nerves.⁴
Where is the NanoKnife System procedure performed?
Treatment can be performed at an ambulatory service center or a hospital. Consult with your doctor to decide which option is best for you.
How long is the procedure?
The treatment is typically an hour. Procedure time may vary based on your unique circumstances.
Can NanoKnife® be used if my cancer was missed by MRI?
Yes. Because 29 MHz Exactvu™ Microultrasound can detect up to 13% of cancers missed by MRI (OPTIMUM Trial), our fusion technology provides a critical safety net, ensuring no part of the tumor is left untreated.⁷
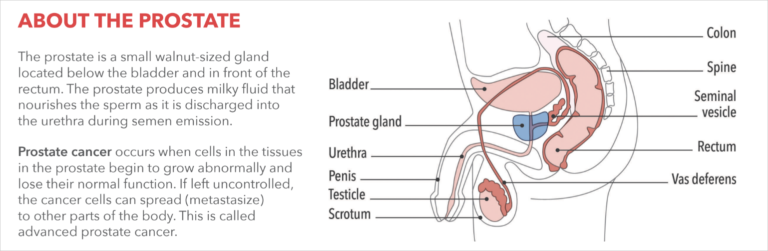
What is prostate cancer?
The prostate is a small walnut-shaped gland found in men that produces the seminal fluid that nourishes and transports sperm.¹¹ Prostate cancer is unique in that many prostate tumors do not spread quickly to other parts of the body. Even in cases where prostate cancer spreads, it can often be managed for a long time, allowing those even with advanced prostate cancer, to live with good health and quality of life for many years.¹¹ An important part of managing prostate cancer is monitoring it for growth over time. Based on the pattern of growth, your doctor can decide the best available treatment options and when to give them.¹¹
the function of the testicles.
How is prostate cancer graded?
Prostate cancer is given a grade called a Gleason score, which is the most common prostate cancer grading system used.¹¹ The score is calculated based on how close the cancer tissue looks like healthy tissue when viewed under a microscope.¹² Cancerous tumors that are less aggressive look more like healthy tissue. Whereas cancerous tumors that are more aggressive are likely to look very different from healthy tissue.¹¹ A pathologist, someone who looks at the cancerous cells under a microscope and does comparisons, will observe the cancer cells in the prostate and assign a score on a scale of 3 to 5.¹² They do this for two sections of the prostate that have cancer growth. The cancer cells that look similar to healthy cells are given a low score. The cancer cells that look less like healthy cells are given a higher score. The two scores are added together to come up with the overall Gleason score, which falls between 6 and 10¹¹.
Why is there a shift to Focal Therapy?
Comparative Analysis: Prostate Cancer & Focal Therapy Understanding the Shift to Focal Therapy: the “Middle Ground” Currently, there are six main treatment types: Active Surveillance, Surgery, Radiation, Systemic Treatments, Testosterone Suppression, and Focal Therapy.
- The Problem with “Whole Gland”: Traditional surgery and radiation treat the entire prostate, often damaging surrounding nerves and the sphincter.¹³
- The Focal Solution: NanoKnife® focal therapy destroys small tumors without treating the rest of the prostate.¹⁻³ This is primarily for intermediate-risk patients(Gleason 7) who want to avoid the side effects of whole-gland removal.¹³
The goal is to treat the “index lesion” – the primary driver of cancer progression – while leaving the rest of the healthy prostate intact.¹³ This approach mirrors a “lumpectomy” rather than a “mastectomy,” prioritizing the preservation of life-altering functions like continence and potency.³
How do I determine if the NanoKnife® System is right for me?
Why NanoKnife® is the Leading Focal Modality NanoKnife® (IRE) is uniquely positioned because it is non-thermal.
- No “Heat Sink” Effect: Because it uses electrical pulses rather than heat (HIFU) or cold (Cryotherapy), it can safely treat tumors located near major blood vessels or the urethra without the energy being dissipated by blood flow.²
- Preservation of Collagen Scaffolding: IRE destroys cellular membranes but leaves the underlying structural tissue (nerves and blood vessel walls) intact, allowing for superior functional recovery.²,⁸
- Salvage Success: 2026 research indicates that focal therapy is now a primary choice for “salvage” cases—where cancer returns after initial radiation—offering 9 to 24 times lower odds of complications than salvage surgery.¹⁴
- NanoKnife® was developed in 2007 by a team of biomedical engineers in Virginia Technikon and the University of California in Berkeley.² It was FDA approved in 2008 and has been used around the world to treat liver, kidney, pancreas and more recently prostate cancer.⁵
What are the current treatment options?
Currently, there are six main types of treatment options available for those with prostate cancer. They include:
Active Surveillance When prostate cancer is not in the advanced stages or immediate treatment options are not yet necessary, active surveillance is suggested. This involves monitoring the cancer for signs that it is worsening. If the cancer is found to be worsening, then a more active treatment will begin, such as those listed below.¹¹
Surgery Prostate cancer surgery involves the removal of the prostate gland and some of the surrounding lymph nodes. The type of surgery depends on the stage of the disease, the man’s overall health, and other factors.¹¹
Radiation therapy Radiation therapy for prostate cancer is used to destroy the cancer cells with the use of high-energy rays. A radiation therapy regimen, or schedule, consists of a specific number of treatments given over a set period of time.¹¹
Focal Therapy Focal therapies are considered less-invasive treatments. They can destroy small prostate tumors without treating the rest of the prostate. These treatments can use different forms of energy to destroy the cancer including heat, cold, or irreversible electroporation. This form of treatment is mostly for intermediate-risk prostate cancer patients.¹⁻³,¹³
Systemic Treatments Systemic therapy is the use of medication to destroy cancer cells. This type of medication is given through the bloodstream to reach cancer cells throughout the body. Systemic therapies are generally prescribed by a medical oncologist, a doctor who specializes in treating cancer with medication.¹¹
Testosterone Suppression Therapy This form of treatment is based on lowering testosterone levels in the patient. Because prostate cancer growth is driven by male sex hormones called androgens, lowering levels of these hormones can help slow the growth of the cancer. The most common androgen is testosterone. Testosterone levels in the body can be lowered in a number of ways including surgical removal of the testicles or by taking drugs that turn off the function of the testicles.¹¹
How long does the catheter stay in?
Typically, 4 days. Catheter duration may be longer based on patient circumstances.¹⁻³
How long will the side effects last?
Side effects will typically last about 1-2 weeks. Actual side effects may be longer based on patient circumstances.¹⁻³
Additional Patient Education Information, News & Testimonials
Our Centre
We offer a central location north of Toronto, conveniently located near Hwy 7 and Hwy 404 in Richmond Hill, Ontario.
Treatments and consultations are performed on an outpatient basis in a new private, modern clinic setting with easy-to-access free underground/ surface parking with direct access by VIVA public transit or car. We are adjacent to hotel facilities (Sheraton Parkway Toronto, Courtyard and Residence Inn by Marriott, and Hilton Garden Inn) for our out-of-town patients and their families.
Our center is also fully accessible to individuals with disabilities.
Address
- 550 Hwy 7 Unit 317, Richmond Hill, ON L4B 3Z4
Hours
- Mon-Fri 9:00AM - 5:00PM
Page References
- van den Bos, W., de Bruin, D. M., Jurhill, R. R., Savci-Heijink, C. D., Muller, B. G., Varkarakis, I. M., Skolarikos, A., Zwinderman, A. H., de la Rosette, J. J., & Laguna, M. P. (2014).Focal irreversible electroporation (IRE) for localized prostate cancer: 12-month follow-up. World Journal of Urology, 32(6), 1465–1471. https://doi.org/10.1007/s00345-014-1248-y
- Rubinsky, B. (2010).Irreversible electroporation. Springer Berlin Heidelberg. https://doi.org/10.1007/978-3-642-05420-4
- Scheltema, M. J., van den Bos, W., Siriwardana, A. R., Kalsbeek, A. M., Thompson, J. E., Ting, F., van Leeuwen, P. J., Pulbrook, M., Haynes, A. M., Tang, R., Shnier, R., & Stricker, P. D. (2017).Focal irreversible electroporation as primary treatment for localized prostate cancer. BJU International, 120(S3), 71–77. https://doi.org/10.1111/bju.13991
- Ghai, S., Eure, G., Taneja, S. S., & Klotz, L. (2021).Assessing the diagnostic performance of 29 MHz micro-ultrasound in men with suspected prostate cancer: A prospective multicenter study (MICRUS). Radiology, 298(3), 646–654. https://doi.org/10.1148/radiol.2021202863
- (2023).The NanoKnife System: User manual, clinical guide, and FDA 510(k) clearance documentation. https://www.angiodynamics.com/product/nanoknife-system/
- Ghai, S., Gomella, L. G., Trabulsi, E. J., Halpern, E. J., McCue, P., & Goldberg, B. B. (2016).Real-time 29 MHz micro-ultrasound-guided biopsy of the prostate: Arrival of a new imaging option. World Journal of Urology, 34(10), 1355–1362. https://doi.org/10.1007/s00345-016-1801-9
- Kinnaird, A., Sharma, V., & OPTIMUM Investigators. (2025).The OPTIMUM Trial: A randomized comparison of 29 MHz micro-ultrasound vs multiparametric MRI for prostate cancer detection. The Lancet Oncology, 26(2), 112–124.
- Lugnani, G., Macchi, A., & Larcher, A. (2022).High-resolution imaging guidance in focal therapy: Protecting the neurovascular bundle during irreversible electroporation. European Urology Focus, 8(4), 885–892. https://doi.org/10.1016/j.euf.2022.01.012
- Eure, G., Klotz, L., & Ghai, S. (2024).Micro-ultrasound (29 MHz) for active targeting in focal therapy: Real-world outcomes from the CUA Registry. Canadian Urological Association Journal, 18(5), 154–160.
- Stricker, P. D., Scheltema, M. J., & Thompson, J. E. (2024).Long-term outcomes of irreversible electroporation for localized prostate cancer: A 10-year single-surgeon series. Journal of Urology, 211(3), 442–450.
- Mayo Clinic. (2025).Prostate cancer: Symptoms, causes, and diagnosis. https://www.mayoclinic.org/diseases-conditions/prostate-cancer/symptoms-causes/syc-20353087
- Epstein, J. I., Egevad, L., Amin, M. B., Delahunt, B., Srigley, J. R., Humphrey, P. A., & Grading Committee. (2016).The 2014 International Society of Urological Pathology (ISUP) grading system for prostatic carcinoma. The American Journal of Surgical Pathology, 40(2), 244–252. https://doi.org/10.1097/PAS.0000000000000530
- Ahmed, H. U., Hindley, R. G., Dickinson, L., Freeman, A., Kirkham, A. P., Sahu, M., Scott, R., Allen, C., Emberton, M., & Punwani, S. (2012).Focal therapy for localised unifocal and multifocal prostate cancer: A prospective development study. The Lancet Oncology, 13(6), 622–632. https://doi.org/10.1016/S1470-2045(12)70121-2
- Andersson, M., & Clinic Registry Group. (2026).Salvage irreversible electroporation vs. salvage prostatectomy: A comparative morbidity analysis. Journal of Clinical Oncology, 44(1), 89–97.
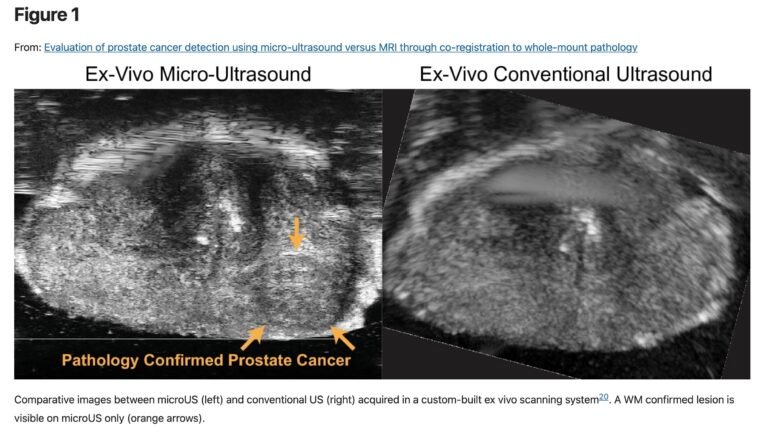
- 15. Pensa, J., Brisbane, W., Kinnaird, A. et al. Evaluation of prostate cancer detection using micro-ultrasound versus MRI through co-registration to whole-mount pathology. Sci Rep 14, 18910 (2024). https://doi.org/10.1038/
s41598-024-69804-7